Nonlinear spatial integration in retinal bipolar cells shapes the encoding of artificial and natural stimuli.
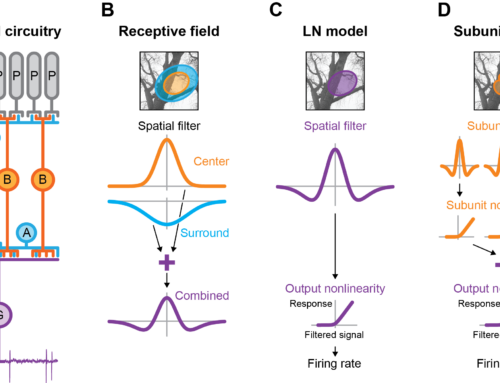
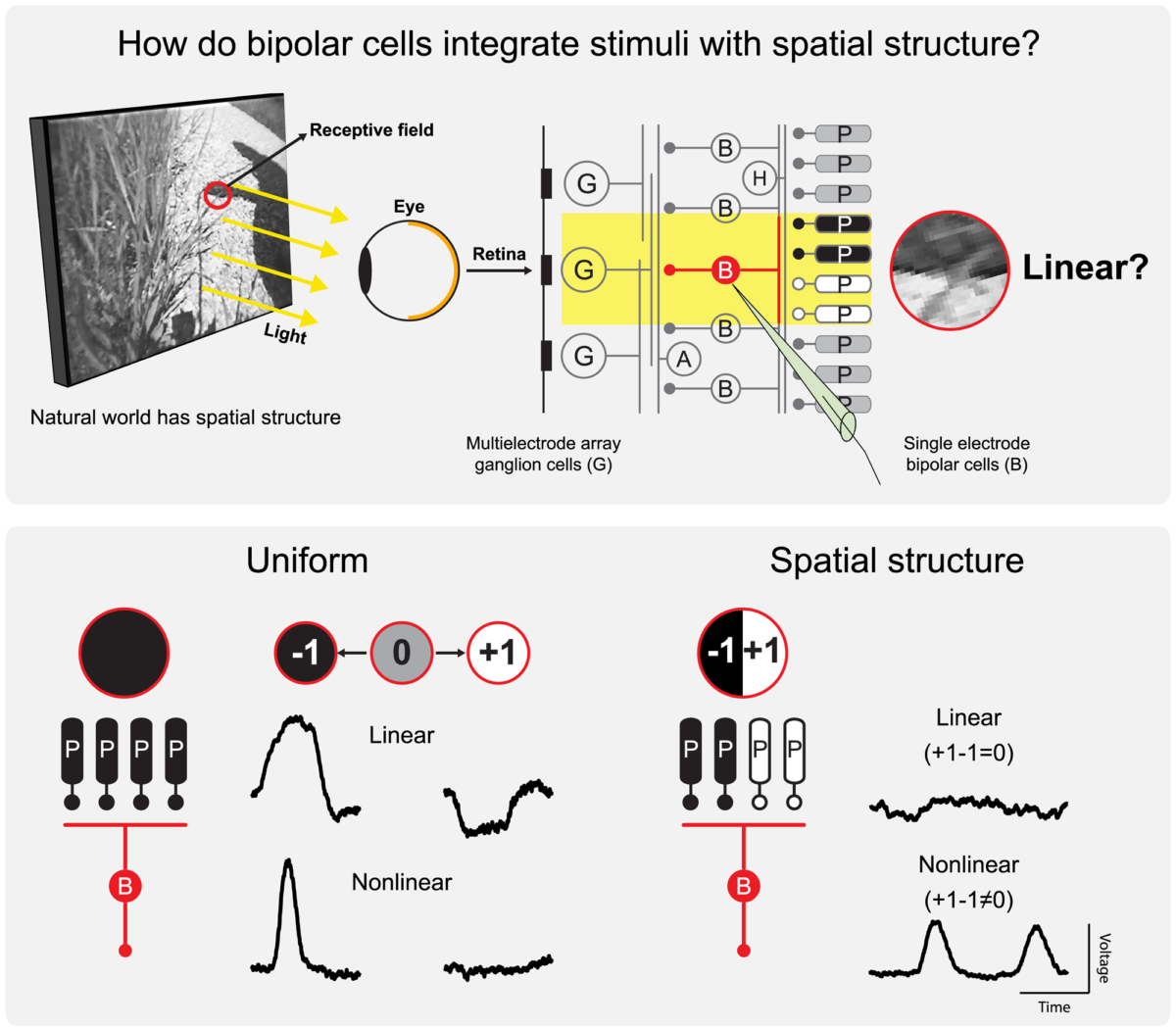
The retina dissects the visual scene into parallel information channels, which extract specific visual features through nonlinear processing. The first nonlinear stage is typically considered to occur at the output of bipolar cells, resulting from nonlinear transmitter release from synaptic terminals. In contrast, we show here that bipolar cells themselves can act as nonlinear processing elements at the level of their somatic membrane potential. Intracellular recordings from bipolar cells in the salamander retina revealed frequent nonlinear integration of visual signals within bipolar cell receptive field centers, affecting the encoding of artificial and natural stimuli. These nonlinearities provide sensitivity to spatial structure below the scale of bipolar cell receptive fields in both bipolar and downstream ganglion cells and appear to arise at the excitatory input into bipolar cells. Thus, our data suggest that nonlinear signal pooling starts earlier than previously thought: that is, at the input stage of bipolar cells.